A) 1.00 V
B) 1.12 V
C) 1.24 V
D) 0.36 V
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An oxidation occurs at the ____ of a Galvanic cell.
A) intersection
B) anode
C) p-n junction
D) cathode
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formation of thin layers of material on the electrodes of a secondary cell will result in:
A) an increase in the EMF of the battery.
B) formation of explosive hydrates.
C) shorter periods of usefulness between recharging sessions.
D) the creation of j-p junction gaps at the electrode surfaces.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Copper has a greater tendency to corrode than does zinc.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The SHE is assigned a voltage of ____ V.
A) − 1.000
B) 1.000
C) 0.150
D) 0.000
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The salt bridge between 1/2 reactions maintains the electrical balance of a Galvanic cell. This bridge is filled with:
A) strong electrolytes.
B) inert carbon.
C) weak electrolytes.
D) interstitial membranes.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the E value for the Galvanic cell formed from these two half-reactions at these concentrations?
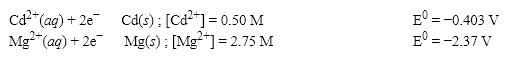
A) 1.90 V
B) 1.95 V
C) 1.99 V
D) 2.20 V
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In balancing electrochemical half-reactions in acidic media, one can assume an excess of both water and OH-.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
All dry cell batteries are rechargeable.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How long will it take to collect 1.0 L of H2 (g) at STP in the electrolysis of 2.0 M HCl with a current of 4.25 A?
A) 15 min
B) 34 min
C) 2.8 hr
D) 11 hr
F) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The species undergoing reduction is referred to as the oxidizing agent.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Electrolysis involves using an external current to drive an electrochemical reaction in a non-spontaneous direction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which mechanism is most energy efficient?
A) internal combustion motor
B) a voltaic fuel cell
C) electricity from coal fired plants
E) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The Nernst equation describes the pH required to convert electrolytic reactions into spontaneous Galvanic cells.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the E0 value for the Galvanic cell formed from these two half-reactions?

A) −1 .43 V
B) +1.43 V
C) −0 .93 V
D) +0.93 V
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Single use, non-rechargeable batteries are referred to as:
A) primary cells
B) secondary cells
C) tertiary cells
D) electrolytic cells
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the correct balanced chemical equation for the cell: 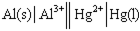
A) Al( s ) + Hg2+( aq ) → Al3+( aq ) + Hg( l )
B) 2 Al( s ) + Hg2+( aq ) → 2 Al3+( aq ) + Hg( l )
C) Al( s ) + 3 Hg2+( aq ) → Al3+( aq ) + 3 Hg( l )
D) 2 Al( s ) + 3 Hg2+( aq ) → 2 Al3+( aq ) + 3 Hg( l )
F) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The kinetics of uniform corrosion will speed up considerably in the presence of good electrical conducting salts such as NaCl
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Electrolysis can be used to electroplate metals onto surfaces.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of Cl in HClO4?
A) 1+
B) 2+
C) 7+
D) 8+
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 40
Related Exams