A) orbit the nucleus in well-defined circular orbits.
B) radiate electromagnetic energy continuously.
C) act as if spread out in a "cloud" around the nucleus.
D) behave as predicted by quantum mechanics.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In the Bohr model of the hydrogen atom both the energy and orbital angular momentum of the electron are quantized.
B) False
Correct Answer

verified
True
Correct Answer
verified
True/False
Photocopiers make use of the photoelectric effect.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
When the speed of an electron is increased the de Broglie wavelength is decreased.
B) False
Correct Answer

verified
True
Correct Answer
verified
Multiple Choice
The person who expounded the uncertainty principle was
A) Max Planck.
B) Albert Einstein.
C) Neils Bohr.
D) Erwin Schrödinger.
E) Werner Heisenberg.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
The peak of the radiation curve of a blackbody moves upward toward higher intensity as its temperature __________.
Correct Answer

verified
Correct Answer
verified
True/False
The energy level diagram for hydrogen 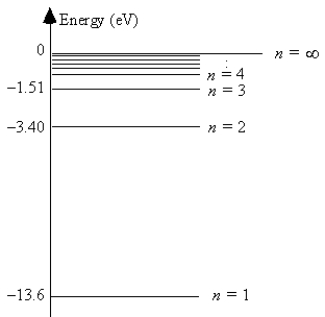 -The ionization energy of hydrogen is 13.6 eV.
-The ionization energy of hydrogen is 13.6 eV.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
When the frequency of the light is increased in a photoelectric effect experiment,the energy of the emitted electrons increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Match each item with the correct statement below. -causes an electron to make a transition to a higher energy level
A) angular momentum
B) de Broglie wavelength
C) emission spectrum
D) energy
E) ground state
F) hologram
G) laser
H) photoelectric effect
I) photon
J) photon absorption
K) photon emission
L) uncertainty principle
M) wave function
O) G) and I)
Correct Answer

verified
Correct Answer
verified
True/False
The characteristic peaks in an x-ray spectrum of an atom occur because of rapid deceleration of the bombarding electrons.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The peak of the radiation curve of a blackbody moves upward toward higher intensity as its temperature increases.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The energy of an oscillating atom in a blackbody can be any value within a certain limited range.
B) False
Correct Answer

verified
False
Correct Answer
verified
Short Answer
Find the energy of a photon whose frequency is 5 × 1012 Hz.
Correct Answer

verified
Correct Answer
verified
True/False
The photoelectric effect occurs when electrons are emitted when light strikes the surface of a metal.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a laser
A) light is amplified by stimulated emission of radiation.
B) the emitted light is monochromatic.
C) the emitted light is coherent.
D) there is a population inversion of electrons in atoms.
E) all of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The chemical composition of stars can be determined from lines in their spectra.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
According to the Pauli Exclusion Principle for n = 4,the occupation limit of electrons is:
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the speed of an electron increases,its de Broglie wavelength
A) increases.
B) decreases.
C) stays the same.
D) may increase or decrease.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A quantum of electromagnetic radiation is a
A) wave function.
B) photon.
C) de Broglie wave.
D) laser.
E) hologram.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lowest energy level in an atom is its
A) ionization level.
B) emission level.
C) absorption level.
D) photon level.
E) ground state.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 143
Related Exams