A) It is used to power yet more cellular work.
B) It is captured to store energy as more ATP.
C) It is used to generate ADP from nucleotide precursors.
D) It is lost to the environment.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
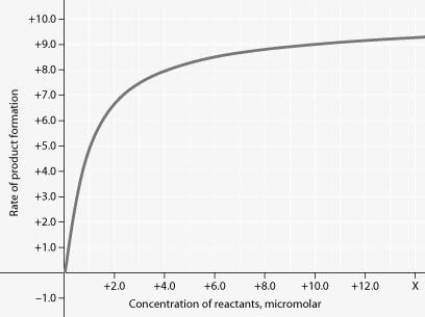 Rate of an enzyme-catalysed reaction as a function of varying reactant concentration, with the concentration of enzyme constant
In the figure, why does the reaction rate plateau at higher reactant concentrations?
Rate of an enzyme-catalysed reaction as a function of varying reactant concentration, with the concentration of enzyme constant
In the figure, why does the reaction rate plateau at higher reactant concentrations?
A) Feedback inhibition by product occurs at high reactant concentrations.
B) Most enzyme molecules are occupied by substrate at high reactant concentrations.
C) The reaction nears equilibrium at high reactant concentrations.
D) The rate of the reverse reaction increases at high reactant concentrations.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Zinc, an essential trace element for most organisms, is present in the active site of the enzyme carboxypeptidase. The zinc most likely functions as ________.
A) a noncompetitive inhibitor of the enzyme
B) an allosteric activator of the enzyme
C) a cofactor necessary for enzyme activity
D) a coenzyme derived from a vitamin
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
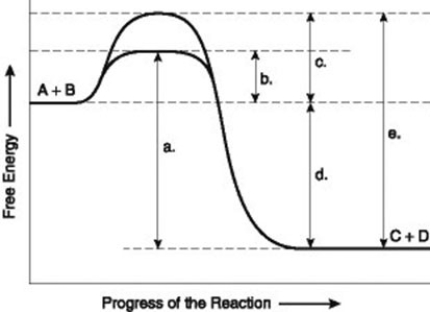 - The figure illustrates the energy states associated with the reaction A + B ↔ C + D. Which of the following in the figure would be the same in either an enzyme-catalysed or a noncatalysed reaction?
- The figure illustrates the energy states associated with the reaction A + B ↔ C + D. Which of the following in the figure would be the same in either an enzyme-catalysed or a noncatalysed reaction?
A) a
B) b
C) c
D) d
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
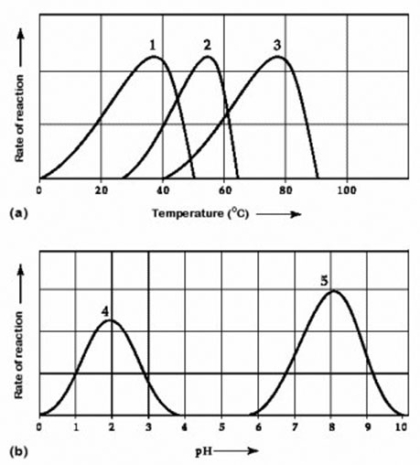 -Activity of various enzymes at various temperatures (a) and at various pH (b)
Which curves on the graphs may represent the temperature and pH profiles of an enzyme taken from a bacterium that lives in a mildly alkaline hot springs at temperatures of 70°C or higher?
-Activity of various enzymes at various temperatures (a) and at various pH (b)
Which curves on the graphs may represent the temperature and pH profiles of an enzyme taken from a bacterium that lives in a mildly alkaline hot springs at temperatures of 70°C or higher?
A) curves 1 and 5
B) curves 2 and 5
C) curves 3 and 4
D) curves 3 and 5
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The relationship between catabolism and anabolism is most similar to the relationship between which of the following pairs of terms?
A) exergonic; spontaneous
B) exergonic; endergonic
C) free energy; entropy
D) work; free energy
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following characteristics is most likely to be associated with an enzyme that catalyses two different chemical reactions?
A) The enzyme contains α-helices and β-pleated sheets.
B) The enzyme is subject to competitive inhibition and allosteric regulation.
C) The enzyme is composed of at least two subunits.
D) Either the enzyme has two distinct active sites or the substrates involved in the two reactions have very similar structures.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A number of systems for pumping ions across membranes are powered by ATP. Such ATP-powered pumps are often called ATPases, although they do not often hydrolyse ATP unless they are simultaneously transporting ions. Because small increases in calcium ions in the cytosol can trigger a number of different intracellular reactions, cells keep the cytosolic calcium concentration quite low under normal conditions, using ATP-powered calcium pumps. For example, muscle cells transport calcium from the cytosol into the membranous system called the sarcoplasmic reticulum (SR) . If a resting muscle cell's cytosol has a free calcium ion concentration of 10-7 while the concentration in the SR is 10-2, then how is the ATPase acting?
A) ATPase activity must be powering an inflow of calcium from the outside of the cell into the SR.
B) ATPase activity must be transferring ![]() i to the SR to enable this to occur.
i to the SR to enable this to occur.
C) ATPase activity must be pumping calcium from the cytosol to the SR against the concentration gradient.
D) ATPase activity must be opening a channel for the calcium ions to diffuse back into the SR along the concentration gradient.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is true of metabolism in its entirety in all organisms?
A) Metabolism depends on a constant supply of energy from food.
B) Metabolism uses all of an organism's resources.
C) Metabolism consists of all the energy transformation reactions in an organism.
D) Metabolism manages the increase of entropy in an organism.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about anabolic pathways is true?
A) They are usually spontaneous chemical reactions.
B) They consume energy to build up polymers from monomers.
C) They release energy by degrading polymers to monomers.
D) They decrease the entropy of the organism and its environment.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements describes a common characteristic of catabolic pathways?
A) They combine small molecules into larger, more energy-rich molecules.
B) They require energy from ATP hydrolysis to break down polymers into monomers.
C) They are endergonic and release energy that can be used for cellular work.
D) They are exergonic and provide energy that can be used to produce ATP from ADP and ![]() i.
i.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
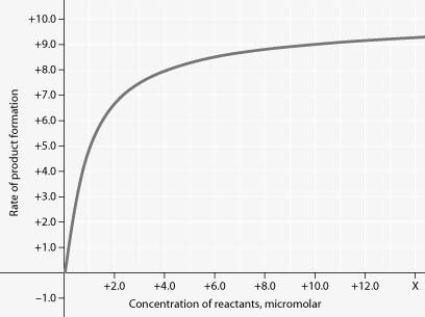 Rate of an enzyme-catalysed reaction as a function of varying reactant concentration, with the concentration of enzyme constant.
For the enzyme-catalysed reaction shown in the figure, if the initial reactant concentration is 1.0 micromolar, which of these treatments will cause the greatest increase in the rate of the reaction?
Rate of an enzyme-catalysed reaction as a function of varying reactant concentration, with the concentration of enzyme constant.
For the enzyme-catalysed reaction shown in the figure, if the initial reactant concentration is 1.0 micromolar, which of these treatments will cause the greatest increase in the rate of the reaction?
A) doubling the activation energy needed
B) cooling the reaction by 10°C
C) doubling the enzyme concentration
D) increasing the concentration of reactants to 10.0 micromolar, while reducing the concentration of enzyme by 1/2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is true for all exergonic reactions?
A) The products have more total energy than the reactants.
B) The reaction proceeds with a net release of free energy.
C) The reaction goes only in a forward direction: all reactants will be converted to products, but no products will be converted to reactants.
D) A net input of energy from the surroundings is required for the reactions to proceed.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is an important consequence of the first law of thermodynamics for a living organism?
A) The energy content of an organism is constant.
B) An organism ultimately must obtain all of the necessary energy for life from its environment.
C) The entropy of an organism decreases with time as the organism grows in complexity.
D) Organisms grow by converting energy into organic matter.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aspects of enzyme structure is best described by a clasping handshake analogy?
A) The specific manner in which an enzyme folds to form secondary and tertiary structures.
B) The specific manner in which an enzyme interacts with water.
C) The specific manner in which an enzyme binds substrate.
D) The specific manner in which an enzyme is denatured by low pH.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the ΔG of ATP hydrolysis in the cell about twice as great as the ΔG of ATP hydrolysis in a test tube under standard conditions?
A) A cell is an open system, whereas a test tube is a closed system.
B) ATP hydrolysis in a test tube releases more heat than ATP hydrolysis associated with cellular metabolism.
C) Reactant and product concentrations in the test tube are different from those in the cell.
D) ATP hydrolysis in cells is catalysed by enzymes, whereas the reaction in a test tube occurs spontaneously.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How might a change of one amino acid at a site, distant from the active site of an enzyme, alter the substrate specificity of an enzyme?
A) by changing the stability of the enzyme
B) by changing the three-dimensional conformation of the enzyme
C) by changing the optimum pH for the enzyme
D) by changing the binding site for a noncompetitive inhibitor
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
 - The figure illustrates the energy states associated with the reaction A + B ↔ C + D. Which of the following represents the activation energy required for the enzyme-catalysed reaction in the figure?
- The figure illustrates the energy states associated with the reaction A + B ↔ C + D. Which of the following represents the activation energy required for the enzyme-catalysed reaction in the figure?
A) a
B) b
C) c
D) d
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a metabolic pathway, succinate dehydrogenase catalyses the conversion of succinate to fumarate. The reaction is inhibited by malonic acid, a substance that resembles succinate but cannot be acted upon by succinate dehydrogenase. Increasing the amount of succinate molecules to those of malonic acid reduces the inhibitory effect of malonic acid. Which of the following statements correctly describes the role played by molecules described in the reaction?
A) Succinate dehydrogenase is the enzyme, and fumarate is the substrate in the reaction.
B) Succinate dehydrogenase is the enzyme, and malonic acid is the substrate in the reaction.
C) Succinate is the substrate, and fumarate is the product in the reaction.
D) Fumarate is the product, and malonic acid is a noncompetitive inhibitor in the reaction.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
________ is a regulatory mechanism in which the end product of a metabolic pathway inhibits an enzyme that catalyses an early step in the pathway.
A) Allosteric inhibition
B) Cooperative inhibition
C) Feedback inhibition
D) Metabolic inhibition
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 67
Related Exams